As a transition metal disulfide (TMD), tin disulfide (SnS2) is an economical and practical electrocatalyst which can be used for hydrogen evolution reaction in alkaline electrolyte. However, the catalytic hydrogen evolution ability of SnS2 has been limited due to its lack of obvious intrinsic catalytic activity and poor adsorption capacity for hydrogen.
In order to improve the catalytic hydrogen evolution capacity of SnS2 catalyst, we reported a method to promote efficient hydrogen evolution by exposing high activity (100) surface on SnS2/SnO2 electrocatalyst. The first author was Wu Jing, PhD candidate of Hunan University.
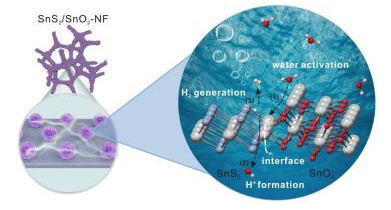
1. The experimental results showed that the SnS2/SnO2 heterostructure was designed and grown on the nickel foam (denoted as SnS2/ sno2-NF), and the SnS2/ sno2-NF catalyst with rich heterogeneous interface showed a highly active crystal surface (100). At such a heterogeneous interface, The hydrolytic energy and adsorption energy of hydrogen atom are optimized to enhance the electron transfer process and make it exhibit excellent hydrogen evolution performance in alkaline electrolyte.
2. The SnS2/SnO2 heterostructure introduces more active (100) surfaces or high-density active sites, accelerates the diffusion kinetics of electrons and ions, reduces the water dissociation energy, and optimizes the adsorption energy of hydrogen atom. On the catalyst, excellent HER performance was achieved in alkaline medium.
3. Under the conditions of overpotential of 108 mV, current density of -10 Ma ·cm-2 and tafel slope of 50.1 mV Dec-1, the catalyst can maintain good catalytic performance of hydrogen evolution in 1 M KOH
Reference:Jing Wu, et al, Exposing highly active (100) facet on a SnS
2/SnO
2 electrocatalyst to boost efficient hydrogen evolution
DOI:
10.1016/j.apcatb.2021.120200